 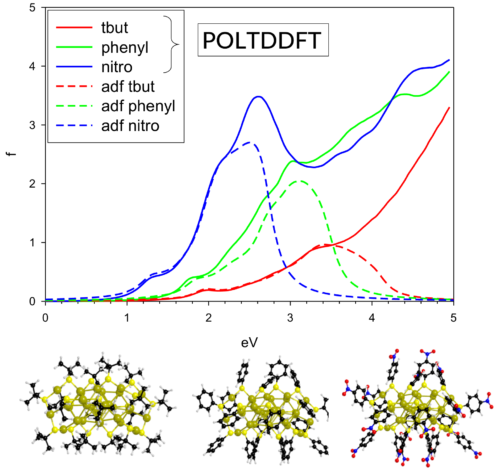
MOFs carry, arguably more than any other type of material, the potential to fulfil crystal engineering, for the cluster and ligand connectivity, and properties, can be to certain extent controlled 17. MOFs are porous materials consisting of metal clusters that are linked to each other by organic bridging ligands. In crystal engineering studies-the design and synthesis of functional crystalline structures based on a bottom-up approach from smaller building blocks-scientists have shown remarkable chemical mechanisms to prelude and govern synthesis of metal–organic framework (MOF) crystals 9, 10, 11, 12, 13, 14, 15, 16. Clock reactions involving phase transitions, in which for instance precipitating or crystallizing particle dimensions oscillate during the course of reaction, have never been shown to exist, despite nonlinear dynamics suspected to govern several important condensation processes, such as the hardening of cement 8. These are solution-driven oscillations in the solid state, but ‘direct’ oscillatory behaviour exists in conversion and temperature during certain polymerization reactions 7. 6 fabricated in this way a device capable of producing 20 mW kg −1 of power. It is possible to use a clock reaction to produce oscillatory pH or temperature trajectories that drive a muscle-like mechanical response from a polymeric material 5. A particularly exciting development is the use of chemical actuation to fabricate a synthetic muscle. Great interest exists in coupling oscillatory clock reactions to processes occurring in the solid state 4. In broad context, clock reactions belong to a class of nonlinear chemical phenomena linked to several natural processes and rhythms. 
This makes it a popular classroom reaction. If starch is added, the oscillations can be followed by corresponding changes in the solution colour. Iodide is not consumed in the overall reaction and serves as autocatalyst. As a result of the difference in reaction rates and the fact concentrations of the autocatalytic redox-pair iodide/iodine are coupled in nonlinear kinetics, the iodine concentration is seen to shoot up after an initial lag time after which it oscillates to equilibrium. The first reaction is slow, the second one fast. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
